Posted by Chrom Tech on 2nd Feb 2026
Using Hydrogen as a Carrier Gas for Gas Chromatography
Hydrogen as a Carrier Gas
Hydrogen is quickly becoming a preferred carrier gas for gas chromatography (GC) due to its efficiency, cost-effectiveness, and renewable availability. As helium supplies become increasingly limited, many laboratories are switching to hydrogen to maintain reliable and sustainable GC operations.
A Renewable Resource
One of hydrogen’s greatest advantages is its abundance and ability to be generated on demand. Using hydrogen generators, laboratories can produce hydrogen through water electrolysis—eliminating the need for gas cylinders and reducing supply dependency. By contrast, helium is a finite byproduct of natural gas extraction and is becoming scarcer as fossil fuel reserves decline. This makes hydrogen a more sustainable, long-term solution for laboratories worldwide.
Lower Operating Costs
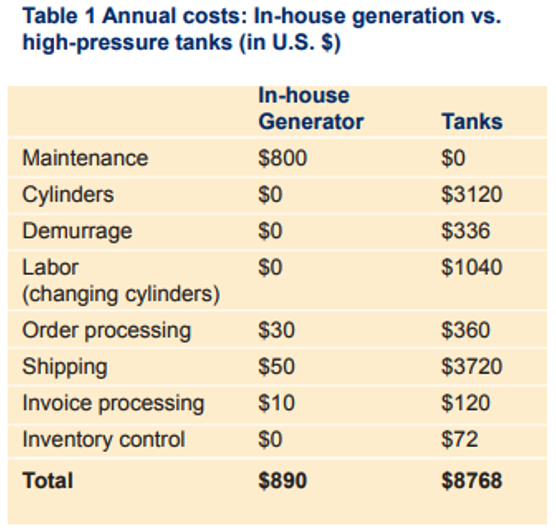
Hydrogen not only offers consistent availability but also significant cost savings over time. While helium prices continue to rise, hydrogen can be generated in-house for a fraction of the cost. A one-time investment in a hydrogen generator provides continuous gas supply, eliminating recurring cylinder rental and delivery expenses.
Higher Efficiency and Faster Chromatography
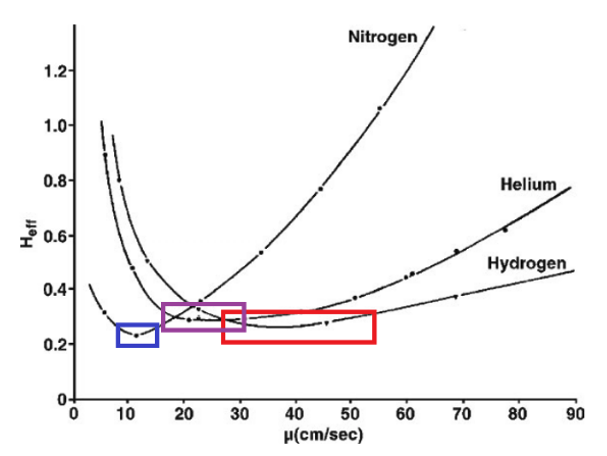
In capillary GC, hydrogen exhibits superior chromatographic efficiency compared to helium. According to the Golay equation (a form of the Van Deemter relationship), hydrogen achieves minimal plate height across a broad linear velocity range (25–55 cm/sec), whereas helium reaches optimal efficiency at approximately 20 cm/sec. This allows hydrogen to achieve similar separation quality at nearly twice the flow rate of helium, enabling faster analyses without sacrificing resolution.
Shorter Run Times and Longer Column Life
Because hydrogen operates efficiently at higher linear velocities, GC methods can be completed in less time. Shorter analysis times reduce oven ramping and temperature exposure, which not only speeds equilibration between runs but also extends column lifetime. This combination of faster throughput and lower maintenance costs makes hydrogen a strong choice for laboratories aiming to increase productivity.
Safety and Practical Considerations
Safety
Despite being flammable, hydrogen is safe to use in GC systems when handled properly. Modern hydrogen generators include built-in leak detection, pressure regulation, and automatic shutoff features, making them safer than gas cylinders. For more information, see our related blog Are Hydrogen Generators Safe?.
Plumbing and System Setup
For best results, deliver high-purity hydrogen to your GC using 316 stainless steel tubing. Stainless steel is durable, non-reactive, and resistant to oxidation—unlike copper, which can degrade over time. Install shutoff valves, inline pressure relief valves (vented to a hood or exhaust), and flashback arrestors for added safety. As with any gas plumbing project, always check for leaks before operation.
Method Conversion
Transitioning from helium to hydrogen is straightforward. You can run hydrogen at the same flow rate as your previous helium methods for similar retention times. To take advantage of hydrogen’s faster efficiency, simply increase your flow rate. Chrom Tech’s technical team can assist with GC method conversion to optimize your new setup.
GC Columns and FID Flame Adjustments
Faster carrier gas flow rates deliver more total gas to the FID (flame ionization detector). When using large-ID columns (e.g., 0.53 mm), reduce hydrogen fuel flow to prevent over-fueling the flame. Alternatively, switch to a smaller GC column ID for better balance between flow rate and detector stability.
Using Hydrogen with GC/MS
Hydrogen is fully compatible with mass spectrometry. Some GC/MS instruments, such as Agilent MSD systems, may require part modifications (e.g., a 6 mm draw-out lens instead of a 3 mm). Ensure your vacuum pump is suitable for hydrogen use—high-efficiency turbo pumps are typically recommended. Always verify specifications with your instrument manufacturer before conversion.
Conclusion
Hydrogen offers laboratories a powerful combination of sustainability, efficiency, and cost savings for gas chromatography. With proper system setup and safety measures, hydrogen provides faster analysis times, longer column life, and a reliable alternative to helium. Chrom Tech supplies a full range of hydrogen generators, tubing, and GC columns to help your lab make the transition with confidence.
Frequently Asked Questions About Using Hydrogen in GC
Is hydrogen safe to use as a carrier gas?
Yes. When used with a hydrogen generator and proper fittings, hydrogen is safe for laboratory use. Generators include safety systems that prevent overpressure and automatically shut down in case of leaks.
Can I use hydrogen with my existing GC methods?
Yes. Hydrogen can typically replace helium at similar flow rates with minimal method changes. You may choose to optimize flow for faster runs and higher efficiency.
Is hydrogen compatible with GC/MS systems?
Hydrogen works well with GC/MS instruments, though some may require part modifications like larger draw-out lenses or upgraded vacuum pumps. Always consult your manufacturer before switching gases.


